PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) is an evidence-based set of guidelines designed to help researchers improve the transparency, quality, and consistency of reporting in systematic reviews and meta-analyses. The guidelines were initially published in 2009 and were later updated in 2020. PRISMA focuses on ensuring that systematic reviews and meta-analyses are reported in a clear and comprehensive manner, allowing readers to assess the reliability and validity of the findings.
PRISMA consists of a 27-item checklist that covers various aspects of the systematic review and meta-analysis process.
These items address areas such as the research question, study selection, data extraction, synthesis, and interpretation of results. The goal is to provide a standard reporting framework that enhances the transparency and reproducibility of systematic reviews, which are often used in evidence-based practices, especially in healthcare.
Key Components of PRISMA
Title and Abstract
The title should clearly indicate that the work is a systematic review or meta-analysis. The abstract should provide a concise summary of the review, including objectives, methods, and key findings. The abstract should be structured, typically with subheadings like “Background,” “Objectives,” “Methods,” “Results,” and “Conclusions” (Liberati et al., 2009).
Rationale
The rationale for the systematic review should be explicitly stated. This typically includes a description of the issue being investigated, its significance, and why a systematic review is appropriate. The rationale helps justify the need for the review in light of existing research (Moher et al., 2009).
Objectives
The objectives should be clearly defined, often in the form of a research question or hypothesis. This helps guide the scope and focus of the review. For example, an objective could be to evaluate the effectiveness of a specific medical intervention for a condition.
Eligibility Criteria
PRISMA requires a detailed explanation of the inclusion and exclusion criteria for selecting studies. This includes specifying study designs (e.g., randomized controlled trials, observational studies), populations, interventions, and outcomes of interest. The purpose is to ensure that only relevant studies are included in the review.
Information Sources
The sources of information (e.g., databases, registers) used for the systematic review should be clearly documented. This includes specifying which databases were searched and the time period during which the search was conducted (Moher et al., 2009). The transparency of search methods allows for the reproducibility of the review.
Study Selection
PRISMA recommends providing a clear description of the process used for selecting studies, including how studies were screened and how decisions about inclusion and exclusion were made. This may involve multiple reviewers to ensure reliability.
Data Extraction
A detailed description of the data extraction process should be included. This includes how data were collected from each study (e.g., outcomes, sample sizes, methodological details) and any adjustments made to account for missing or incomplete data.
Risk of Bias
The potential risk of bias in individual studies should be assessed and reported. This can involve using standardized tools to evaluate the risk of bias in the included studies, such as the Cochrane Risk of Bias tool. Recognizing bias helps assess the credibility and generalizability of the findings.
Synthesis of Results
The results of the included studies should be synthesized in a transparent and systematic manner. If a meta-analysis is conducted, statistical methods for combining results should be clearly outlined. For narrative syntheses, the approach to summarizing findings should be described in detail.
Subgroup and Sensitivity Analyses
If applicable, PRISMA encourages authors to report subgroup analyses (e.g., based on age, gender) and sensitivity analyses (e.g., testing the robustness of results by excluding certain studies). These analyses provide deeper insight into the factors that might influence the results.
Discussion
The discussion should provide an interpretation of the findings, including their implications for practice or policy, limitations of the review, and any potential sources of bias (Moher et al., 2009). It should also highlight areas where future research is needed.
Funding
PRISMA guidelines include a requirement to disclose the funding sources for the systematic review. This ensures transparency and helps readers assess potential conflicts of interest that may influence the review process or interpretation of results.
PRISMA Flow Diagram
A key feature of the PRISMA guidelines is the PRISMA Flow Diagram, which provides a visual representation of the study selection process. The diagram helps readers understand the number of studies identified, screened, assessed for eligibility, and ultimately included in the review. It also helps document reasons for exclusion, which enhances transparency (Moher et al., 2009).
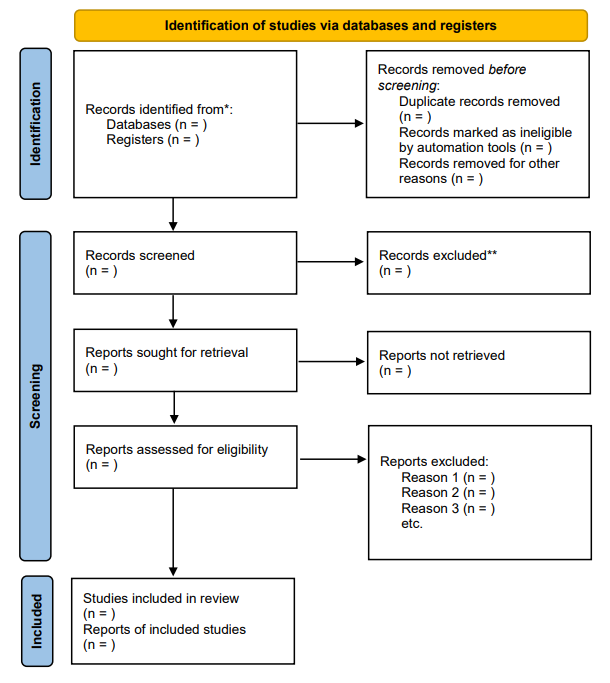
Article Selection Procedure – PRISMA Flow Diagram (Page et al., 2021)
PRISMA 2020 Update
In 2020, PRISMA was updated to reflect changes in systematic review practices, emerging methodologies, and advancements in reporting standards. The revised guidelines incorporate new items that address the growing importance of registering protocols, considering environmental factors, and managing complex data types. Some of the key changes in the 2020 update include:
- Enhanced focus on the protocol for the systematic review (e.g., pre-registration of reviews).
- More explicit reporting on statistical methods, including how data were analyzed and any software used for meta-analysis.
- Greater emphasis on conflict of interest and transparency in funding sources.
- Encouragement for tailored reporting for different types of systematic reviews, including those that assess interventions, diagnostics, and prognostic factors (Page et al., 2021).
Importance of PRISMA in Systematic Reviews
The PRISMA guidelines are important because they enhance the quality and reliability of systematic reviews and meta-analyses, which play a critical role in informing evidence-based practice, particularly in fields like healthcare, education, and social sciences. By providing clear and standardized reporting, PRISMA helps reduce bias, improves reproducibility, and ensures that systematic reviews can be critically appraised and used in decision-making.
Conclusion
PRISMA provides a comprehensive framework for reporting systematic reviews and meta-analyses. By following these guidelines, researchers can improve the clarity, transparency, and reproducibility of their work, which is crucial for advancing scientific knowledge and informing evidence-based decisions. The 2020 update of PRISMA reflects the evolving landscape of research and ensures that reviews meet modern standards of rigor and transparency.
References
- Liberati, A., Altman, D. G., Tetzlaff, J., Mulrow, C., Gøtzsche, P. C., Ioannidis, J. P., … & Moher, D. (2009). The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLOS Med, 6(7), e1000100. https://doi.org/10.1371/journal.pmed.1000100
- Moher, D., Liberati, A., Tetzlaff, J., Altman, D. G., & The PRISMA Group. (2009). Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLOS Med, 6(7), e1000097. https://doi.org/10.1371/journal.pmed.1000097
- Page, M. J., McKenzie, J. E., Bossuyt, P. M., Boutron, I., Hoffmann, T. C., Mulrow, C. D., … & Moher, D. (2021). The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. PLOS Med, 18(3), e1003583. https://doi.org/10.1371/journal.pmed.1003583

Leave a Reply